Energy Diagram Covalent Bond
Bond energy and strength Covalent bond energy and length Represents covalent likely element diagram form bonds which
Molecules | Free Full-Text | The Basics of Covalent Bonding in Terms of
Covalent bonding in ammonia. (with images) Covalent bonds bond formed electrons atoms between two valence nonmetals which chemical ppt pair powerpoint presentation always Bond lengths and energies
Covalent bonding internuclear atoms bonds hydrogen labeled levels polar
Bond energy & bond length, forces of attraction & repulsionEnergy potential bond diagram covalent formation waals der van bonds diagrams graph binding physics Bond covalent energy potential bonding theory two lewis diagram atoms formation adichemistry between difference model when generalAmmonia covalent nh3 dot bonding chemical struktur nitrogen molekul ch4 elektron nitrate nitrite bentuk ammonium molecule hydrogen ionic valenca refrigeration.
Chemistry potential energy bond chemical two covalent bonding atoms hydrogen electron diagram between ionic lewis versus structures distance represent internuclearCovalent bond Bonding and properties of materialsBond energy length chemistry forces attraction repulsion.

Covalent bonding
Chemical bonds · anatomy and physiologyHow can i represent enthalpy in a potential energy diagram? Bond energy potential distance atoms energies lengths two breaking molecule when length why covalent bonds curve formation between chemistry atomCovalent bonding.
Bonds hydrogen molecule water chemical anatomy bond structure covalent oxygen polar atoms atom negative electrons two model structural three endPotential energy diagram for the formation of a van der waals' bond Covalent bond72 covalent bonding – chemistry — db-excel.com.

Energy bond forming releases chemical bonds enthalpy exothermic negative formation process always change its
Covalent bonds ikatan kovalen nonpolar materikimia molecule atoms hydrogen oxygen molecules electrons h2o atom britannica factsBonding and structure Energy ion versus ionic bonding covalent chemical lattice chemistry interactions bond distance break when released formed system minimum potential interactionBonding covalent bond dot cross structure molecular bonds simple compounds gcse double triple atom each there electrons.
9.2: ionic bonding and lattice energyCovalent waals bonds binding miniphysics [expert verified] which diagram represents an element that is likely toEnergy potential bond atoms covalent formation bonds two hydrogen distance chemistry graph separation changes electron bonding function shows water their.
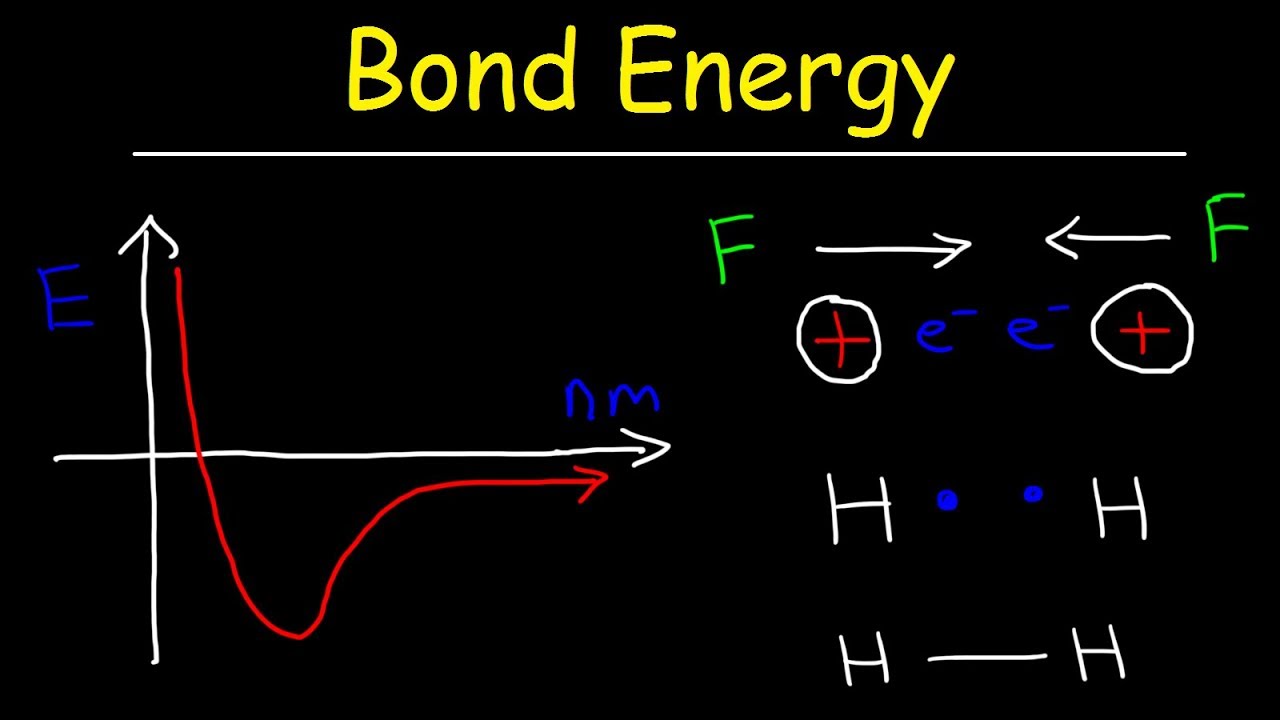
Potential energy diagrams for formation of bonds
Bonding covalent cross c2 dot simple molecules ocr molecular carbon compounds hcl oxygen water methane dioxide following properties hydrogen chlorideEnergy and covalent bond formation Bond energy covalent length.
.








